 
This is the reason why H is always a terminal atom and never a central atom. Hydrogen only needs to form one bond to complete a duet of electrons. Atom (Group number)īecause hydrogen only needs two electrons to fill its valence shell, it follows the duet rule. Table showing 4 different atoms, each of their number of bonds, and each of their number of lone pairs. 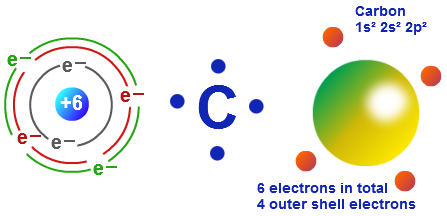
Effective nuclear charge increases from left to right across the periodic table because of the increasing atomic. In each case, the sum of the number of bonds and the number of lone pairs is 4, which is equivalent to eight (octet) electrons. This decreases the pull on valence electrons towards. Group 1A (or IA) of the periodic table are the alkali metals: hydrogen (H), lithium (Li), sodium (Na), potassium (K), rubidium (Rb), cesium (Cs), and francium (Fr).These are (except for hydrogen) soft, shiny, low-melting, highly reactive metals, which tarnish when exposed to air. The number of electrons required to obtain an octet determines the number of covalent bonds an atom can form. Oxygen and other atoms in group 16 obtain an octet by forming two covalent bonds: Electrical Conductivity: Value given for graphite. Discovery Year: Known since ancient times. Boiling Point: Value given for diamond form. For example, four hydrogen atoms can bond with a carbon atom, which has four valence electrons, to form methane. This means it can bond with many elements. To obtain an octet, these atoms form three covalent bonds, as in NH 3 (ammonia). Click any property name to see plots of that property for all the elements. Hydrogens valence number is one, because it has only one valence electron and needs only one shared electron to fill its energy levels. 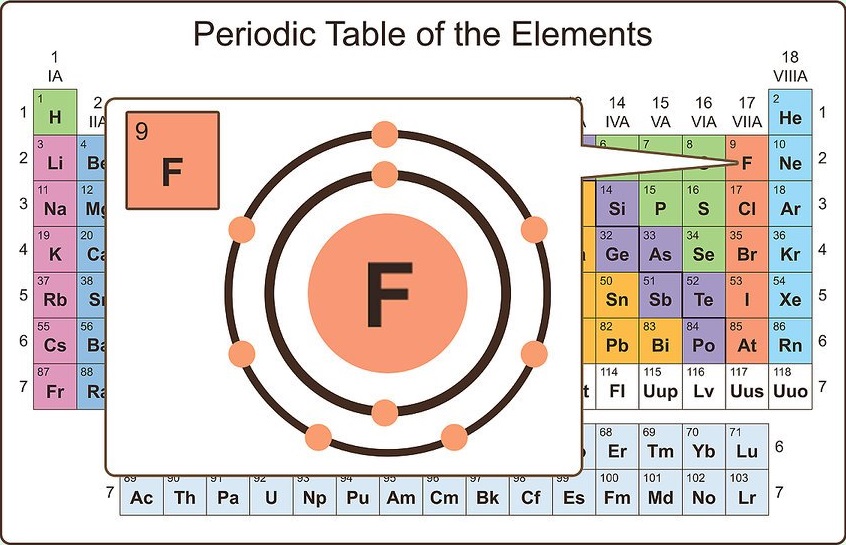
On the periodic table, it is the first of six elements in column (group) 14, which have in common the composition of their outer electron shell. Group 15 elements such as nitrogen have five valence electrons in the atomic Lewis symbol: one lone pair and three unpaired electrons. Carbon (from Latin:carbo 'coal') is a chemical element with symbol C and atomic number 6. The transition elements and inner transition elements also do not follow the octet rule since they have d and f electrons involved in their valence shells. Because hydrogen only needs two electrons to fill its valence shell, it is an exception to the octet rule and only needs to form one bond. These four electrons can be gained by forming four covalent bonds, as illustrated here for carbon in CCl 4 (carbon tetrachloride) and silicon in SiH 4 (silane). For example, each atom of a group 14 element has four electrons in its outermost shell and therefore requires four more electrons to reach an octet. The number of bonds that an atom can form can often be predicted from the number of electrons needed to reach an octet (eight valence electrons) this is especially true of the nonmetals of the second period of the periodic table (C, N, O, and F). How Many Covalent Bonds Are Formed The number of bonds that an atom can form can often be predicted from the number of electrons needed to reach an octet (eight valence electrons) this is especially true of the nonmetals of the second period of the periodic table (C, N, O, and F). \): 3D molecule examples. From left to right: water molecule, ammonia molecule, and methane molecule 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
